Chromatography Is Used to Do Which of the Following
Chromatography is a physical method that is used to separate complex mixtures. High Performance Liquid Chromatography a technique in analytical chemistry used to separate identify and quantify each component in a mixture.

Separation Of A Mixture Worksheet And Lab Collection Matter Unit Separation Paper Chromatography
The mixture of different components is flushed through the system at different rates.

. -compound is placed on stationary phase. C Determine molecular weight distribution of polymers. Column Chromatography is used to isolate active ingredients.
If above their boiling points which of the following is a good choice. This will separate complex mixtures of chemicals or proteins into their various different. The separation of a mixture into its components is a physical process.
It is very helpful in separating compound mixtures. Retention time can be used to identify a compound in a mixture using gas chromatography. CHE 231 Final- Column Chromatography.
The term chromatography is derived from Greek chroma meaning colour and graphein meaning to write. It is based on the different speed of displacement of the various components of the mixture on a porous medium paper under the action of a moving solvent. 1 Electrolysis 2 Chromatography.
Which one of the following methods can be used to obtain highly pure metal which is liquid at room temperature. In which type of chromatography the stationary phase held in a narrow tube and the mobile phase is forced through it under pressure. That is because the components of the mixture are not chemically combined they can be separated by physical means.
-mobile phase solubilizes the components. Chromatography can test blood alcohol drug purity food purity and essential oil quality. D Measure the number average molecular weight of polymers.
Following a reaction for 22 min. Thin layer chromatography can be used to identify natural products like essential oils or volatile oil fixed oil glycosides waxes alkaloids etc. It is widely used in separating multicomponent pharmaceutical formulations.
The solvent is called the mobile phase. Chromatography employing a gas as the moving carrier medium. Why does it elute in this order.
It is used to determine drug estimation from drug formulations. Industries often use it to monitor processes to test for contamination or ensure a process is going as planned. That is the line in which you will spot your mixtures to separate.
PDF Download - The use of native cation-exchange chromatography to study aggregation and phase separation of monoclonal antibodies. Chromatography separates the components of a mixture by their distinctive attraction to the mobile phase and the stationary phase. Usually a thin layer chromatography plate is around 57 cm high and a line is drawn around 0510 cm from the bottom.
Ferrocene elutes first because it is less polar than acetylferrocene when hexane is used as the mobile phase. -mobile phase passed through the stationary phase. Chromatography is vital for any chemical research because a chemical reaction rarely gives us 100.
The solvent used for chromatography will be selected based on the polarity of the substances in the mixture you want to separate. Chromatography is a method for separating mixtures based on differences in the speed at which they migrate over or through a stationary phase. Following 2 days of storage on ice from the fact that agitation-induced aggregation the suspension was centrifuged at 0 C to collect the shows little correlation with thermodynamic stabi- precipitate.
Types of Column Chromatography. Which one of the following will not affect the retention time of a compound in a gas chromatography column. Low pressure liquid chromatography.
This could be charge polarity or a combination of these traits and pH balance. Ideally the components of a sample should have different boiling points. It is either used to test how pure something is or is used as a technique to purify something from a mixture.
Introduction Chromatography has two main uses. What of the following chromatography techniques would be best to purify a protein from alysate samplea. It is important that you spot the mixtures above.
It is used to purify of any sample and direct comparison is done between the sample and the authentic sample. Chromatography Problem Set Go over the concepts of partition coefficient retention time dead time capacity factor relative retention factor. The porous medium is called the fixed phase.
TLC on silica stained with permanganate. Chromatography is useful as a tool to determine -the absence or presence of components of mixtures whose identities are known Chromatography is more commonly used to -separate a mixture before individual components are analyzed by other techniques such as mass spectrometry Adsorption chromatography. One type of chromatography that can be used is paper chromatography.
Use 1 for the first and 4 for the last. It is used to remove impurities. Chromatography is a method of separating the constituents of a solution based on one or more of its chemical properties.
GC may be used on either organic or inorganic analytes but the sample must be volatile. Column chromatography is used in this experiment to separate the compounds in the mixture from each other. Chromatography is the technique for the separation purification and testing of compounds.
In this process we apply the mixture to be separated on a stationary phase solid or liquid and a pure solvent such as water or any gas is allowed to move slowly over the. High performance liquid chromatography. Chromatography is a way to look at complex mixtures by separating them into their components.
Following the procedure for this experiment which compound will be eluted from the column first ferrocene or acetylferrocene. In essence the solution is passed through a medium which will hinder the movement of some particles more than others. Suggest the order you would expect the following to elute from the column.
Size exclusion chromatography can be used to do the following analysis except a Separate and identify proteins. B Determine the tertiary structure of proteins. Used to isolate metabolites from biological fluids.

Agitated Glass Nutsche Filters Chemical Industry Manufacturing Filters
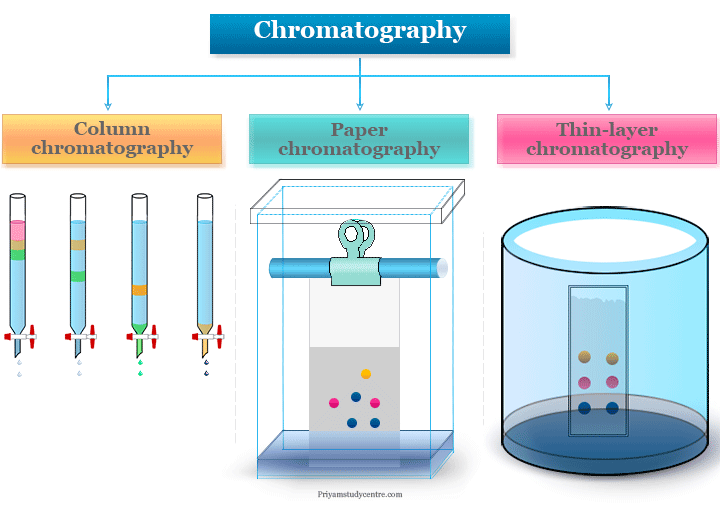
Chromatography Techniques Definition Principle Types

How To Select The Headspace Vial By Temperature Gas Chromatography Vials Headspace

Labbench Prentice Science Biology

Hplc High Perpromance Liquid Chromatography Or High Pressure Liquid High Pressure History Definition Pressure

Hplc High Perpromance Liquid Chromatography Or High Pressure Liquid History Definition High Pressure Column

Chromatography Shalom Education

Stardand Autosampler Vial For Gc And Hplc System Vials Manufacturing Screw

6 Easy Biology Science Experiments For Kids Science Experiments Kids Science Experiments Biology Experiments

Thin Layer Chromatography Tlc Is A Very Commonly Used Technique In Synthetic Chemistry For Id Paper Chromatography Thin Layer Chromatography Science Projects

The Analyzer Source Gas Chromatography Ads Detection

Chromatography Biology Units Chemistry Biology Labs

Mcat Organic Chemistry Question Organic Chemistry Questions Organic Chemistry Mcat
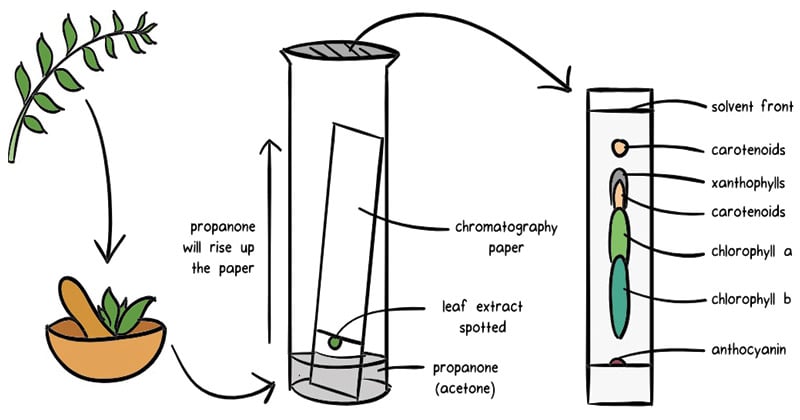
Chromatography Definition Principle Types Applications

Principles Of Chromatography Stationary Phase Article Khan Academy
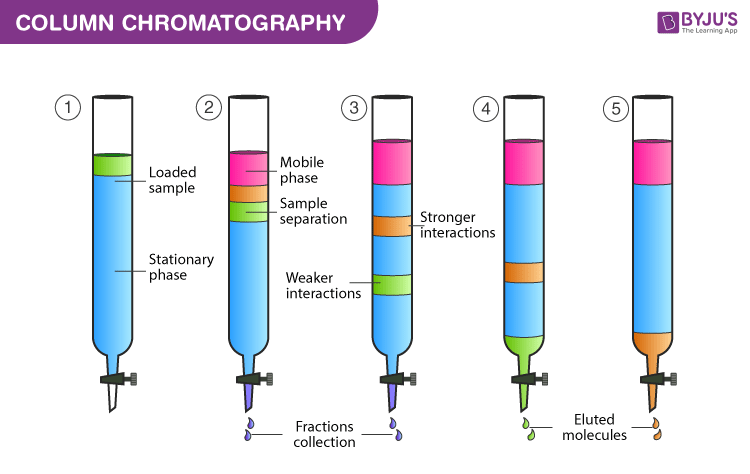
Column Chromatography Principle Procedure Applications Elution In Chromatography

Pengertian Sublimasi Dan Kromatografi Lengkap Dengan Macam Jenisnya Chemistry Classroom Teaching Chemistry Science Chemistry

Pin On Gcse Chemistry Revision Notes

Classification Of Matter Quiz Physics Classroom Matter Unit Chemical Changes
Comments
Post a Comment